Introduction
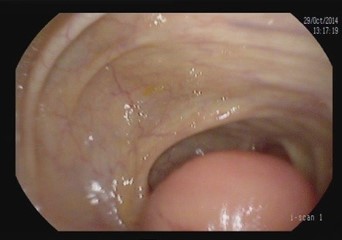
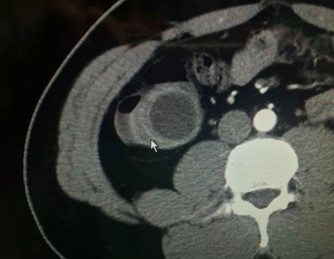
Appendiceal Mucinous Neoplasm (AMN) is a rare and complex tumor of the appendix that’s made up of mucin-producing cells [1,2]. It is described by the World Health Organization (WHO) as three main categories of mucinous neoplasms: Mucinous adenoma, Low-grade Appendiceal Mucinous Neoplasm (LAMN) and Appendiceal carcinoma. Histologic differentiation is important, as well-differentiated tumors have a better prognosis [3,4]. In 2016, the consensus established by the members of the Peritoneal Surface Oncology Group International (PSOGI) suggested to classify mucinous neoplasms of the appendix as serrated polyps, low grade appendiceal mucinous neoplasm (LAMN), High grade Appendiceal Mucinous Neoplasm (HAMN) and mucinous carcinoma. Despite LAMNs are by definition considered low grade, well differenciated tumors, appendiceal perforation by the tumor can results in cellular mucinous tumor deposits distributed throughout the peritoneum known, as pseudomyxoma peritonei (PMP) [3]. When pseudomyxoma peritonei occurs, it is associated with high morbidity and mortality [2-4]. Frequently, appendiceal neoplasm is misdiagnosed because most patients are asymptomatic [5]. The incidence of appendiceal tumors in appendectomy specimens is between 0.9 and 1.7% with, approximately, one half of them being LAMN [6]. These tumors are characterized by the production of mucin, leading to the formation of a mucocele, which can presents with a wide range of clinical manifestations, from asymptomatic incidental findings to acute abdominal pain mimicking appendicitis [6-8]. It is generally found during surgery because of the difficulty of preoperative diagnosis. Thus, imaging is an important tool in diagnosis and management. Computerized Tomography (CT) is particularly useful when identifying tumors in the ileocecal region, as it offers multi-planar, high-definition anatomic evaluation of the intra-abdominal contents.
Patients and methods
This study aimed at assessing the diverse surgical strategies required in the management of patients with AMN. A retrospective review of a prospectively mantained database identied 18 patients with AMN who underwent surgery, between September 2005 and December 2024. Seven patients with Pseudomyxoma Peritonei (PMP) at the time of diagnosis and 4 patients with mucinous carcinoma. LAMN were found in 13 cases. Twelve of 18 patients were female (66.7%) and the median age was 59 years (range: 42–84). Preoperative diagnosis was acute appendicitis in 4 patients, and in the rest, it was appendiceal tumor or right lower abdominal mass.
Tumor markers (CEA and CA 19-9) were within normal range at the initial diagnosis, except in 2 cases with carcinoma. Tumor pathological features like tumor size, surgical margins, existence of cellular atypia and stage, as well as postoperative short and longterm outcomes were assessed.
Results
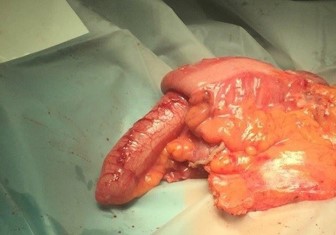
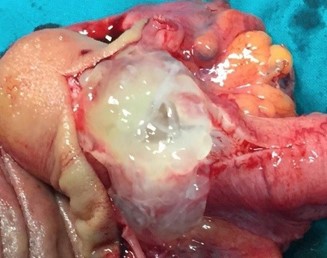
Patients were grouped as patients treated only with appendectomy (5 patients) and as patients treated with cecal resection or Right Hemicolectomy (RH) (13 patients). Patients clinical features, surgical details and oncological outcomes are provided in Table 1. In pathology specimen, median size of primary tumour was 4.5 cm and surgical margins after colonic resection were free of tumor in all cases. Median follow-up time was 25 months (range 5-24 months). Follow-up surveillance was based on of thoracoabdominal computed tomography scan and serum tumor markers every 6 months for 5 years after surgery, and yearly thereafter. Five patients had recurrence. In this series, the majority of patients were female, that is consistent with the literature. As reported by other studies, the patients were usually diagnosed in their 6th or 7th decades. Also, none of patients with LAMN had positive surgical margins; this is also suggestive for watch and waits strategy with appendectomy only. In this series there were 5 patients with T4 tumor. Presenting symptoms included abdominal or pelvic pain (n=9, 50%) and abdominal distention/bloating (n=6, 33%). Two patients had an abdominal mass and 1 was found to have HAMN after surgery, including 1 patient who had a mucocele noted on preoperative screening colonoscopy. Information on abdominal imaging was available for all 18 patients and showed a suspicious appendiceal dilatation in 5 cases.
Table 1: Patient clinical characteristics, surgical details and postoperative outcomes.
| Sex F/M |
Surgery |
Markers (CEA- CA19.9) |
Histology |
Follow up (mons) |
Recurrence Yes/No (Y/N) |
Dead/Alive (D/A) |
| F |
Laparosc append |
Normal |
LAMN |
110 |
N |
A |
| F |
Hemi dx+Hipec |
Normal |
LAMN+PMP |
77 |
N |
A |
| F |
Hemi dx+cytoreduct |
Normal |
LAMN+PMP |
24 |
N |
A |
| F |
Hemi dx |
Pathol. |
Carc. mucin |
108 |
N |
A |
| F |
Open appendec+cytoreduction |
Normal |
LAMN+PMP |
60 |
Y |
A |
| M |
Laparosc append |
Normal |
LAMN |
24 |
N |
A |
| M |
Open appendec |
Normal |
LAMN |
119 |
N |
D |
| M |
Open appendec |
Normal |
LAMN |
204 |
N |
A |
| F |
Hemi dx |
Normal |
Carc. mucin |
180 |
Y |
D |
| M |
Cecal resection+Hipec |
Normal |
LAMN+PMP |
63 |
Y |
D |
| F |
Cecal resection+Hipec |
Normal |
LAMN |
13 |
N |
A |
| F |
Cecal resection+Hipec |
Normal |
Carc. mucin |
48 |
Y |
A |
| M |
Hemi dx+Hipec |
Normal |
HAMN |
32 |
N |
A |
| F |
Hemi dx |
Pathol. |
Carc.mucin+PMP |
61 |
N |
D |
| F |
Laparosc append |
Normal |
LAMN |
27 |
N |
A |
| M |
Cecal resection+Hipec |
Normal |
LAMN+PMP |
32 |
N |
A |
| F |
Hemi dx |
Normal |
LAMN |
29 |
N |
A |
| F |
Hemi dx+Hipec |
Normal |
LAMN+PMP |
33 |
Y |
A |
Table 2: Patient clinical characteristics, surgical details and postoperative outcomes.
| Author |
N. Pats |
Diagnosis |
Median follow up (mons) |
SV/RFS 5 years / 10 years |
| Aguirre et al. [14] |
147 |
Carcinoma |
53 |
91% (5 ys SV) |
| Rauverdink et al. [13] |
121 |
LAMN (78 pts) |
|
166 mons (median SV) |
| HAMN (8 pts) |
60 |
| Carcinoma (35pts) |
| Wang et al. [11] |
159 |
LAMN |
38 |
80% (10 ys SV) |
| Levine et al. [15] |
121 |
LAMN (82pts) |
NS |
51,9% (10 ys SV) |
| HAMN (39pts) |
47,8% (10 ys RFS) |
| Ruff et al. [8] |
995 |
LAMN |
NS |
40% (10 ys SV) |
| Gibson et al. [3] |
216 |
Various type+ PMP |
NS |
96 mons (median SV) |
| Dartigues et al. [12] |
443 |
LAMN (246pts) |
|
73,7 mons (median SV)* |
| HAMN (34pts) |
|
|
| Carcinoma (163pts) |
30.6 |
42,7 mons (median RFS)* |
| Kohler et al. [21] |
612 |
LAMN |
60 |
79,52% (5 ys SV) |
SV: Survival; RFS: Recurrence Free Survival; *: Patients with Carcinoma.
Discussion
Appendiceal mucinous neoplasm accounts for 8-10% of appendiceal tumors and nearly 58% of malignant appendiceal tumors. LAMN is a borderline or low-grade malignant tumor, regardless of rupture, and is characterized by implantation metastasis [2-4]. Its pathogenesis involves atypical hyperplasia of the glandular appendix epithelium that obstructs the appendix with a gradual accumulation of mucus resulting in increased pressure. Mucus penetrates the muscularis mucosa and produces mucinous masses around the appendix and in the retroperitoneum [9-12]. Most cases are asymptomatic, and in the absence of acute infection, the symptoms and signs of LAMN resemble those of chronic appendicitis [13]. In our series, we observed and grouped the cases as patients treated only with appendectomy (5 patients) and as patients treated with cecal resection or Right Hemicolectomy (RH) (13 patients). It is important to underline that, in our experience, some patients underwent more extensive surgery for advanced disease. In fact, we performed surgery with peritoneal cytoreduction and Hipec in 7 cases. As other Authors reported in their analysis, we had similar data at pathology specimen as median size of primary tumour was 4.5 cm and surgical margins after colonic resection were free of tumor in all cases. A correct diagnosis of appendiceal neoplasm may be difficult, but on physical examination large tumors can be found as a complete oval mass with no surrounding adhesions [10]. An appendiceal mucinous cyst that forms from an appendiceal lesion can easily be misdiagnosed as a common appendiceal abscess or right adnexal cyst. In terms of resectability criteria for appendiceal mucoceles, the decision to perform an appendectomy versus a more extensive procedure, such as right hemicolectomy, is guided by several key factors [14-16,21]. Studies have shown that tumors confined to the distal appendix without cecal involvement can be managed with a simple appendectomy, while those extending to the appendiceal base or demonstrating high-grade features require a cecal resection or right hemicolectomy to achieve clear margins and reduce recurrence [5,16]. Additional evidence indicates that involvement of the mesoappendix or adjacent structures increases the risk of mucin spillage and subsequent pseudomyxoma peritonei, warranting more aggressive surgical intervention [6,11,20]. These criteria, along with tumor size and the possibility of lymphatic spread, although rare in lowgrade lesions, are central to intraoperative decision-making. In the study of Rauwerdink et al. [13] it is observed that AMN histology does not always reflects its associated PMP grade, while PMP grade strongly influences survival. By carefully assessing these factors, surgeons can determine the most appropriate extent of resection and thereby optimize patient outcomes. Abdominal ultrasound and CT or MRI are auxiliary diagnostic methods that can distinguish appendiceal mucinous cysts from other lesions before surgery [18,19]. CT is a more accurate imaging method for the diagnosis of appendiceal neoplasms, and shows a cystic mass closely adjacent to the cecum, with a round or long tubular shape, thin wall, and a smooth and regular outline. When CT shows an irregular cyst wall and thick soft tissue, it is more likely to be malignant. MRI performed with a variety of sequences and scanning methods, and high tissue resolution can clearly and consistently distinguish the wall and fluid of the appendiceal mucinous cyst, and more accurately show the integrity and boundary of the cyst wall. In this case, additional MRI evaluation showed typical manifestations of appendiceal cysts [16,19]. Recently in a clinical study, Guaglio et al. [5] observed that if LAMN had been radically resected with appendectomy, even with limited peritoneal spread, it recurred unlikely; furthermore, appendix wall perforation and the presence of mucin, cells, or both outside the appendix did not carry a higher risk of metachronous peritoneal dissemination. Moreover, in the study of Young et al. [6] were analyzed patients with positive surgical margin or perforated neoplasm with mucin on the serosal surface of the appendix, but right hemicolectomy seemed to offer no additional benefit over appendectomy alone. However, Wang et al. [11] operated 159 patients with LAMN and, after a median follow-up time of 38 months, observed that 7 patients recurred and developed pseudomyxoma peritonei. These Authors stated that surgery plus Hipec was necessary for LAMN after complete removal in patients with a high risk of recurrence. At the same, Gibson et al. [3] studied 223 patients with pseudomyxoma peritonei and stated that survival was more closely associated with the grade of the peritoneal disease than with the grade of the primary neoplasm. Their findings support the developing concept that mutational analysis may provide prognostic information in patients with PMP. But, when mucin is found intraoperatively, surgeons should consider appendectomy with possible conversion to an open approach for thorough exploration. Emergency HIPEC is unnecessary during appendectomy; instead, it should be planned electively after resolution of the acute clinical situation and with the definitive histological examination. It is important to underline that in a recent study of Salminen et al. [10] the appendiceal tumor rate in patients with periappendicular abscess was 14.3%, which was higher compared with uncomplicated acute appendicitis (14.3% vs 1.5%) and complicated acute appendicitis without periappendicular abscess (14.3% vs 2.4%). Of these 54 tumors, there were 21 low-grade appendiceal mucinous neoplasms, 20 adenocarcinomas, 8 adenomas, and 5 neuroendocrine tumors. The factors contributing to the development of PMP from appendiceal neoplasm remain unclear. However, based on the results of the most recent analysis, patients with mucin on mesoappendix or neoplastic epithelium extending into muscularis propria/serosa, perforation and positive margin have a higher risk of developing into PMP [11]. Therefore, the majority of the surgeons suggested and performed cytoreduction plus Hipec on these patients.
Conclusions
Appendiceal mucinous neoplasms are complex tumors of the appendix. Histologic type is one of the most significant prognostic factors. However, appendiceal neoplasm is frequently misdiagnosed because most patients are asymptomatic. Iatrogenic rupture of the tumor should be avoided during surgery to reduce the risk of implantation, metastasis and disease recurrence. Spontaneous rupture of the tumor and the spread of mucus to the abdominal cavity can be observed during laparoscopic exploration, indicating that the patient might have a relatively high risk of disease recurrence. Although surgery and peritoneal lavage is performed, and a negative incision margin is obtained, postoperative follow-up should be continued for a long time. The physical examination should be done carefully, and the laboratory and imaging examination results should be comprehensively analyzed to reduce the possibility of misdiagnosis and to ensure prompt diagnosis and treatment, and to improve patient prognosis. Surgery has a critical role in the management of appendiceal mucinous neoplasms. Postoperative management should involve critical review of the pathology report, considering referral to a multidisciplinary team for higher-stage tumors, and implementing long-term surveillance protocols due to the risk of recurrence.
Conflict of interest: The authors declare that they have no conflict of interest.
References
- N Oravec, L Mack, D Hallock, C McClurg, ML Quan: A scoping review of postoperative surveillance strategies for localized, low-grade appendiceal mucinous neoplasms. Am J Sur. 2025; 242: 116202.
- B Turan, AN Sanli, S Acar. Analysis of survival and prognostic factors in appendix adenocarcinoma and mucinous carcinoma. Updates in Surgery. 2025; 77: 327–332.
- J Gibson, RJ Pengelly, A Mirandari, K Boukas, et al. Targeted genetic sequencing analysis of 223 cases of pseudomyxoma peritonei treated by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy shows survival related to GNAS and KRAS status. Cancer Medicine. 2024; 13: e70340.
- M Louis, B Gibson, J Chambers. Optimal surgical approaches for incidentally discovered ruptured mucinous appendiceal neoplasms. Cureus. 2024; 16: e70808.
- M Guaglio, S Sinukumar, S Kusamura, M Milione, F Pietrantonio, L Battaglia, et al. Clinical surveillance after macroscopically complete surgery for low-grade appendiceal mucinous neoplasms (LAMN) with or without limited peritoneal spread: long-term results in a prospective series. Ann Surg Oncol. 2018; 25: 878-884.
- S Young, SK Sueda, M Hotta, ML Sung, VV OʼConnor, AM Leung. Surgical management of appendiceal mucinous neoplasm: is appendectomy sufficient? J Surg Oncol. 2020; 122: 1173-1178.
- C Ayala, A Sathe, X Bai, SM. Grimes, J Shen, et al. Distinct gene signatures define the epithelial cell features of mucinous appendiceal neoplasms and pseudomyxoma metastases. Front Genet. 2025; 16: 1536982.
- SM Ruff, G Kasumova, M Palavalli, OS Eng, L Lambert, C Clarke, et al. Long term outcomes associated with the use of perioperative systemic chemotherapy on low grade appendiceal mucinous neoplasms with pseudomyxoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Front Oncol. 2025; 14: 1456920.
- J Doll, K Maurus, F Kohler, N Matthes, JF Lock, CT Germer, et al. Molecular profiling of low-grade appendiceal mucinous neoplasms (LAMN). Genes, Chromosomes and Cancer. 2024; 63: e23270.
- R Salminen, J Alajaaski, T Rautio, S Hurme, P Nordstrom, E Makarainen, et al. Appendiceal tumor prevalence in patients with periappendicular abscess. JAMA Surg. 2025.
- C Wang, Y Jia, G Shi, L An, X Fan, Zhang, et al. Prophylactic cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for low-grade appendiceal mucinous tumors with early and limited disease after completely removed. Medicine. 2024; 103: 47.
- P Dartigues, V Kepenekian, C Illac-Vauquelin, V Verriele, J Fontaine, et al. Insights into the clinical prognosis of high-grade appendiceal mucinous neoplasms. Am J Surg Pathol. 2025; 49: 499–507.
- P Rauwerdink, D Al-Toma, ECE Wassenaar, MG Raicu, MM Laclé, et al. Reclassification of appendiceal mucinous neoplasms and associated pseudomyxoma peritonei according to the peritoneal surface oncology group international consensus: clinicopathological reflections of a two-center. Cohort Study. Ann Surg Oncol. 2024; 31: 8572–8584.
- N Aguirre, SK Chung, MB Foote, J Shia, E Vakiani, et al. Predictors of recurrence in nonmetastatic appendiceal epithelial cancers: an updated single-center experience over 25 years. Ann Surg Oncol. 2025; 32: 695–702.
- EA Levine, H Cos, KI Votanopoulos, P Shen, G Russell, P Mansfield, et al. Ten-year outcome of a randomized trial: cytoreduction and HIPEC with mitomycin C versus oxaliplatin for appendiceal neoplasm with peritoneal dissemination. Ann Surg Oncol. 2025; 32: 679–686.
- WL Shaib, R Assi, A Shamseddine, OB Alese, C Staley, B Memis, et al.: Appendiceal mucinous neoplasms: diagnosis and management. Oncologist. 2017; 22: 1107–1116.
- AH Omari, MR Khammash, GR Qasaimeh, AK Shammari, MK Yaseen, SK Hammori: Acute appendicitis in the elderly: risk factors for perforation. World J Emerg Surg. 2014; 9: 6.
- PJ Pickhardt, AD Levy, CA Rohrmann, AI Kende: Primary neoplasms of the appendix: radiologic spectrum of disease with pathologic correlation. Radiographics. 2003; 23: 645–662.
- SH Tirumani, M Fraser-Hill, R Auer, W Shabana, C Walsh, F Lee F, et al. Mucinous neoplasms of the appendix: a current comprehensive clinicopathologic and imaging review. Cancer Imaging. 2013; 13: 14–25.
- N Grabill, M Louis, JW Ray, A Tucker, T Walker, J Chambers. Incidental appendiceal mucocele discovery: A case series and literature review Int J Surg Case Rep. 2025; 131: 111281.
- F Kohler, L Reese, A Hendricks, et al. Low-grade mucinous neoplasms (LAMN) of the appendix in Germany between 2011 and 2018: a nationwide analysis based on data provided by the German Center for Cancer Registry Data (ZfKD) at the Robert Koch Institute (RKI). Langenbeck’s Arch Surg. 2022; 407: 3615–3622.